 
It can be defined as the capability or the force with which an atom of an element pulls the shared electrons of a bond towards itself making the sharing as unequal. The molecules which are highly symmetrical are Non-Polar (like BF3).įor more detailed information, you must also check out the article written on BF3 Lewis Structure, Molecular Geometry, and Hybridization.Įlectronegativity is denoted by symbol χ (Greek letter Chi). unsymmetrical then the molecule acquires a polarity (like in NH3 where the Nitrogen atom has the lone pair). If these lone pairs of electrons remain unbalanced i.e. The Lewis structure of the molecule gives us an overview of all the lone pairs of electrons that are present in the molecule. VSEPR (Valence Shell Electron Pair Repulsion) Theory helps to identify the geometrical shape of the molecule which gives us the bond angle. As the bond angle between the bonds determines if the resultant dipole moment of the overall molecule is equal to 0 (Zero) or not. The polarity of a molecule is very much dependent on its structure and geometry. These factors are explained below in detail To find out whether a given molecule is polar or non-polar, we must also understand the factors which are responsible for the polarity of a molecule. Now, we know that the Non-Polarity of BF3 (Boron Trifluoride) is due to its highly symmetrical Trigonal Planar geometry, as predicted by the VSEPR (Valence Shell Electron Pair Repulsion) Theory, which other molecules, like H2O, doesn’t have. Thus, making the molecule BF3 (Boron Trifluoride) as Non-Polar. The three bonds of B-F in BF3 (Boron Trifluoride) are polar (as discussed above) and thus they have a bond dipole moment as shown below:īut the highly symmetrical structure and geometry of BF3 (Boron Trifluoride), as discussed earlier, allows the bond dipole moments to be canceled such that the resultant Molecular Dipole Moment is equal to 0 (Zero). The bond polarity doesn’t always result in overall molecular polarity. However, we must keep in mind that bond polarity is different from overall molecule polarity. It means that the electrons are not shared equally by the two atoms (B and F) instead they are pulled towards F (Fluorine). Thus, each B-F bond in BF3 (Boron Trifluoride) molecule is polar. The difference between the electronegativity values of B and F is equal to 1.94 (3.98 – 2.04 = 1.94) which is greater than 0.5. To determine if a bond is polar or not we must find out the difference between the electronegativity values of the two atoms sharing a bond. The electronegativity of B (Boron) is 2.04 and of F (Fluorine) is 3.98 (maximum) according to the Pauling scale which means that F (Fluorine) will pull the shared electrons towards itself and will thus acquire a partial negative charge (δ-) and B (Boron) will have a partial positive charge (δ+). In BF3 (Boron Trifluoride) molecule there are three bonds of B-F, as shown clearly in Lewis diagram above. We can see that each F (Fluorine) atom has 3 lone pairs of electrons thus the molecular structure is balanced and symmetric.Īccording to the VSEPR (Valence Shell Electron Pair Repulsion) Theory, the BF3 (Boron Trifluoride) molecule has a Trigonal planar geometry with each F-B-F bond angle equal to 120 degrees which again adds to the balance of the molecule making it highly symmetric, as shown:Įlectronegativity & Bond Polarity of BF3 (Boron Trifluoride) 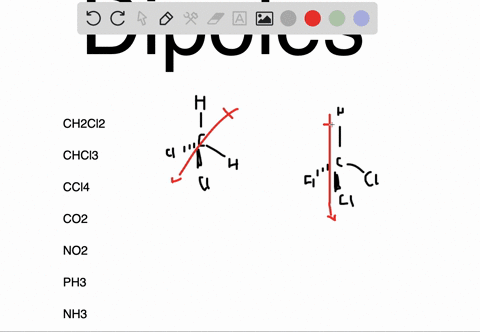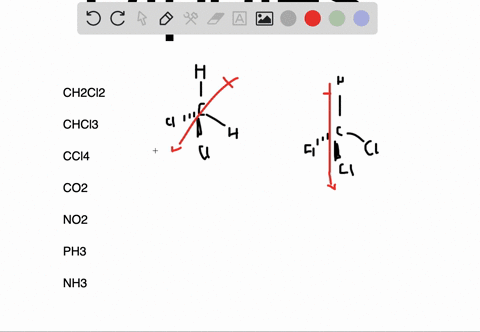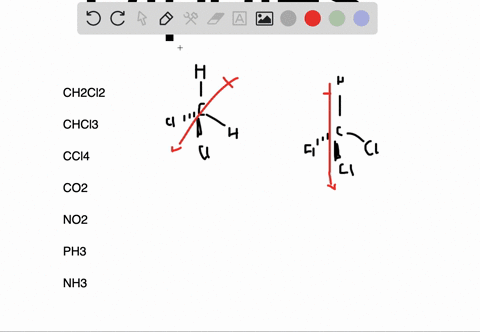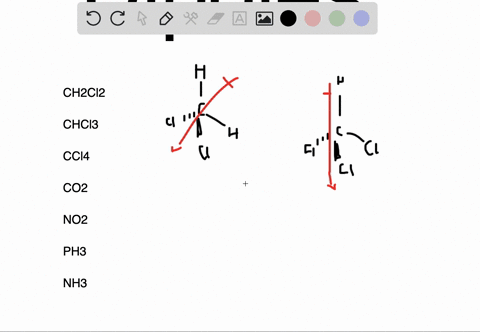 
The valency of B (Boron) is 3 and of F (Fluorine) is 7, thus the Lewis structure of BF3 can be drawn as shown in the figure: The BF3 (Boron Trifluoride) molecule has 1 atom of B (Boron, atomic no: 5) and 3 atoms of F (Fluorine, atomic no: 9). So in this case we do not have a contribution to the molecule's dipole moment from the electrons in the spherically symmetric orbital.Uses of BF3 (Boron Trifluoride) Molecular Structure of BF3 (Boron Trifluoride) It has no directionality, it doesn't "point" in any specific direction because it is spherically symmetric. Let's look at the molecule $\ce$ orbital is spherically symmetric. Let's see if we can use the following picture to help clarify things. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
